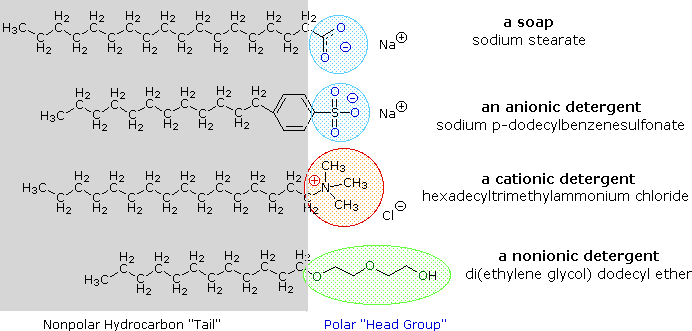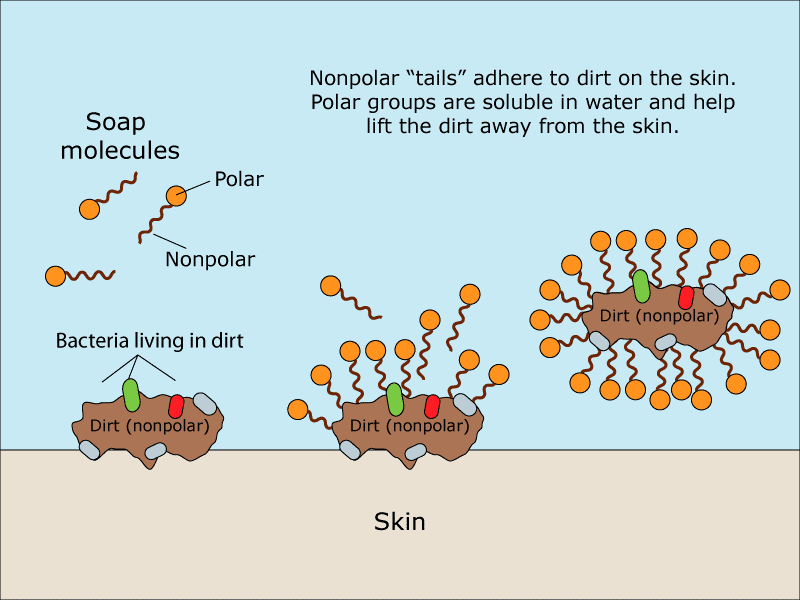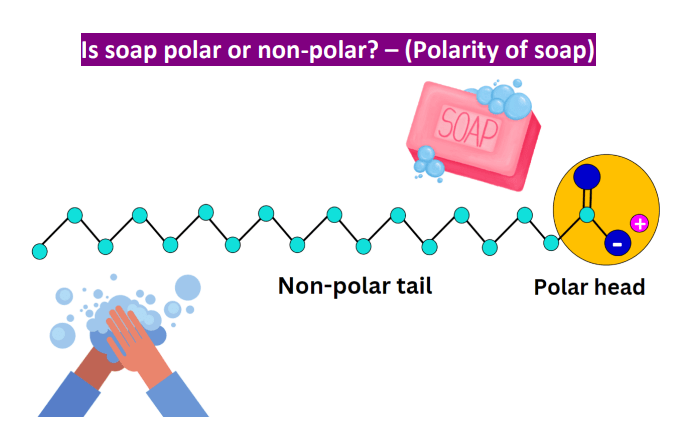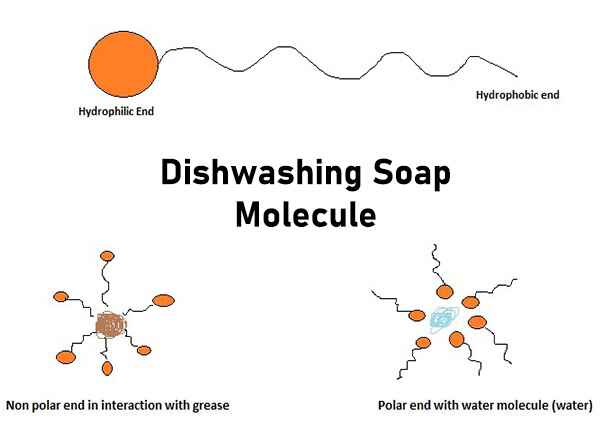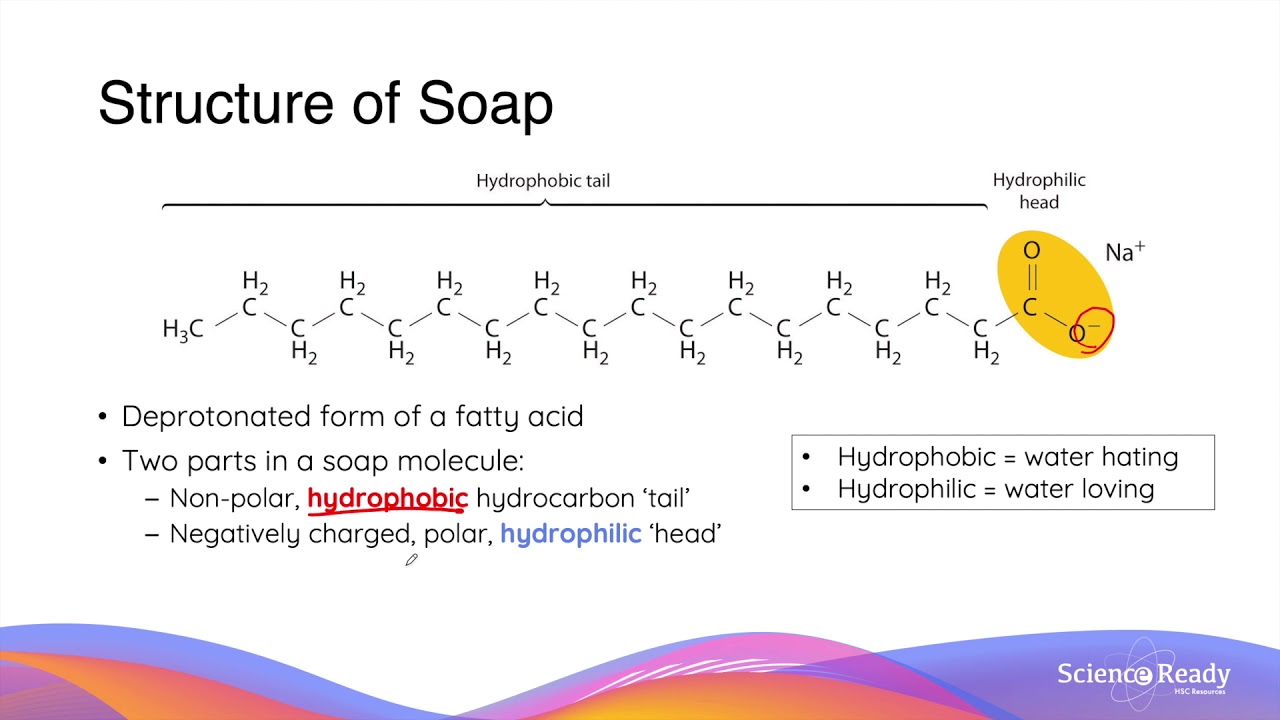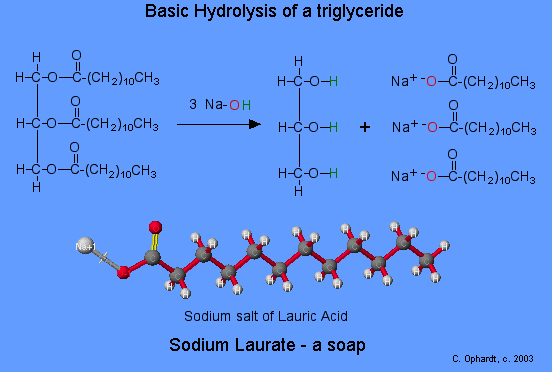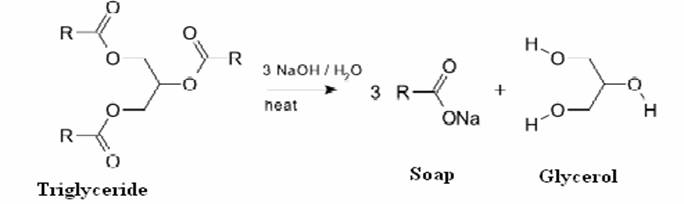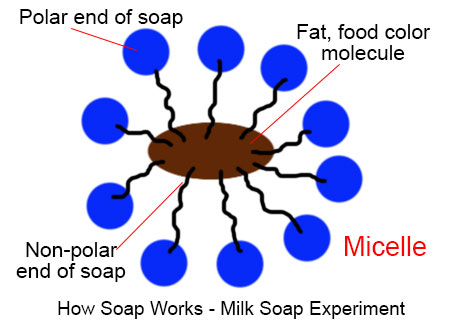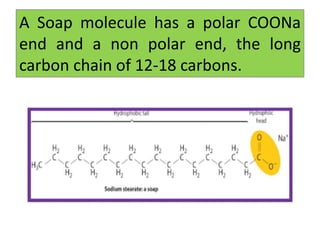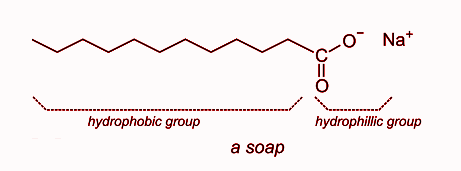
a) What is soap? (b) Describe the structure of a soap molecule with the help of a diagram. (c) Explain the cleansing action of soap. Draw a diagram to illustrate your answer.

Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?